Mdsap Auditing Organizations
If you are searching about Risk Management Auditing in Ann Arbor; MI ⋆ Quality Systems Compliance you've visit to the right place. We have 9 Images about Risk Management Auditing in Ann Arbor; MI ⋆ Quality Systems Compliance like Medical Device Single Audit Program (MDSAP) Audit & Certification | SGS USA; NSF on path to become MDSAP Auditing Organization | Medical Design and and also Risk Management Auditing in Chicago; IL ⋆ Quality Systems Compliance. Read more:
Risk Management Auditing In Ann Arbor; MI ⋆ Quality Systems Compliance
 qscompliance.com
qscompliance.com auditing
Risk Management Auditing In Chicago; IL ⋆ Quality Systems Compliance
auditing risk management chicago
Supplier Management For Medical Device Manufacturers - QSIT And MDSAP
 www.complianceonline.com
www.complianceonline.com supplier mdsap manufacturers device medical management audit cycle classification fda requirements
Medical Device Single Audit Program (MDSAP) Audit & Certification | SGS USA
 www.sgsgroup.us.com
www.sgsgroup.us.com NSF On Path To Become MDSAP Auditing Organization | Medical Design And
 www.medicaldesignandoutsourcing.com
www.medicaldesignandoutsourcing.com nsf mdsap auditing organization certification path international medical medicaldesignandoutsourcing
Global Regulatory Outlook: 2017 And Beyond - OMTEC 2017
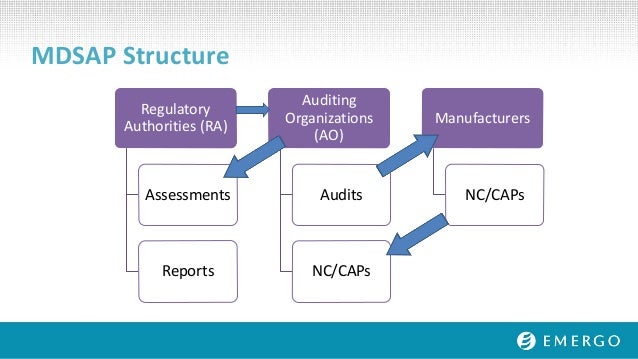 www.slideshare.net
www.slideshare.net regulatory omtec mdsap
MDSAP Consulting -Quality And Regulatory Experts - EMMA International
 emmainternational.com
emmainternational.com mdsap audit
MDSAP - Medical Device Single Audit Program - TSQuality.ch
 tsquality.ch
tsquality.ch mdsap tsquality
MDSAP â€" Medical Device Single Audit Program â€" TSQ Middle East & Asia
mdsap audit tsq tsquality opting
Risk management auditing in chicago; il ⋆ quality systems compliance. Mdsap audit. Nsf on path to become mdsap auditing organization
Komentar
Posting Komentar